Glaucoma
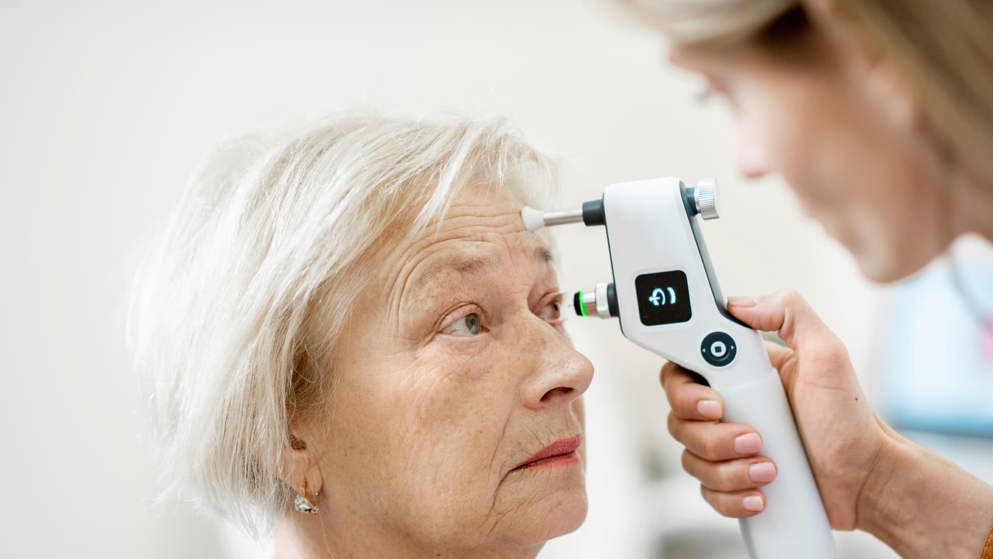
Glaucoma is a group of progressive eye diseases that damage the optic nerve, typically due to increased intraocular pressure (IOP). The most common types include primary open-angle glaucoma and angle-closure glaucoma, each with distinct pathophysiological features. IOP rises when aqueous humor accumulates in the anterior chamber, typically due to impaired drainage. As pressure builds, it compresses the optic nerve, gradually leading to vision loss. Although glaucoma is a leading cause of blindness in adults over 60, timely diagnosis and management can preserve vision.
What is the prevalence of glaucoma?
Globally, glaucoma affects approximately 80 million people, and almost half remain undiagnosed. By 2040, it is projected to cause blindness in around 22 million people. These figures highlight the urgent need for improved screening and awareness.
What symptoms are commonly associated with glaucoma?
Most forms of glaucoma develop silently, without noticeable symptoms. Many patients are diagnosed during routine eye examinations. However, in some cases – such as angle-closure glaucoma – symptoms may appear suddenly and include intense eye pain, redness, blurred vision, halos, headache, and nausea or vomiting.
What are the causes of glaucoma?
While the precise etiology of common glaucoma remains unclear, elevated IOP is a key risk-factor. Lowering IOP remains the cornerstone of treatment and has been shown to slow disease progression.
What treatment options are available for glaucoma?
Glaucomatous damage is irreversible, but early diagnosis, consistent monitoring, and timely treatment can significantly slow or prevent further vision loss. Treatment options include topical eye drops, oral medicines, laser therapy, surgery, or a combination of these approaches. Emerging therapies, such as neuroprotective agents, are also being explored.
Developed by EPG Health for Medthority, independently of any sponsor.
of interest
are looking at
saved
next event
Related news and insights
References
- Soh Z, Yu M, Betzler BK, Majithia S, Thakur S, Tham YC, et al. The Global Extent of Undetected Glaucoma in Adults: A Systematic Review and Meta-analysis. Ophthalmology. 2021;128(10):1393-404.
- Shamsher E, Davis BM, Yap TE, Guo L, Cordeiro MF. Neuroprotection in glaucoma: old concepts, new ideas. Expert Review of Ophthalmology. 2019;14(2):101-13.
- Burton MJ, Ramke J, Marques AP, Bourne RRA, Congdon N, Jones I, et al. The Lancet Global Health Commission on Global Eye Health: vision beyond 2020. The Lancet Global Health. 2021;9(4):e489-e551.
- Nuzzi R, Tridico F. Glaucoma: Biological trabecular and neuroretinal pathology with perspectives of therapy innovation and preventive diagnosis. Front Neurosci. 2017;11:494.
- Cursiefen C, Cordeiro F, Cunha-Vaz J, Wheeler-Schilling T, Scholl HPN. Unmet Needs in Ophthalmology: A European Vision Institute-Consensus Roadmap 2019–2025. Ophthalmic Res. 2019;62(3):123-33.
- Chan MPY, Broadway DC, Khawaja AP, Yip JLY, Garway-Heath DF, Burr JM, et al. Glaucoma and intraocular pressure in EPIC-Norfolk Eye Study: cross sectional study. BMJ. 2017;358:j3889.
- Bonomi L, Marchini G, Marraffa M, Morbio R. The relationship between intraocular pressure and glaucoma in a defined population. Data from the Egna-Neumarkt Glaucoma Study. Ophthalmologica Journal international d'ophtalmologie International journal of ophthalmology Zeitschrift fur Augenheilkunde. 2001;215(1):34-8.
- Wang W, He M, Li Z, Huang W. Epidemiological variations and trends in health burden of glaucoma worldwide. Acta Ophthalmol (Copenh). 2019;97(3):e349-55.
- European Glaucoma Society Terminology and Guidelines for Glaucoma, 5th Edition. Br J Ophthalmol. 2021;105(Suppl 1):1-169.
- Zhang N, Wang J, Li Y, Jiang B. Prevalence of primary open angle glaucoma in the last 20 years: a meta-analysis and systematic review. Sci Rep. 2021;11(1):1-12.
- Tham YC, Li X, Wong TY, Quigley HA, Aung T, Cheng CY. Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology. 2014;121(11):2081-90.
- Stein JD, Khawaja AP, Weizer JS. Glaucoma in Adults—Screening, Diagnosis, and Management: A Review. JAMA. 2021;325(2):164-74.
- Kusuhara S, Nakamura M. Ripasudil Hydrochloride Hydrate in the Treatment of Glaucoma: Safety, Efficacy, and Patient Selection. Clin Ophthalmol. 2020;14:1229-36.
- Quigley HA. 21st century glaucoma care. Eye (Lond). 2019;33(2):254-60.