Bowel disorders
Bowel disorders encompass a spectrum of gastrointestinal (GI) conditions affecting intestinal structure or function, often resulting in digestive symptoms. Major categories include irritable bowel syndrome (IBS) and inflammatory bowel disease (IBD).
How are IBS and IBD characterized?
IBS is a functional GI disorder characterized by recurrent abdominal pain associated with defecation or changes in stool frequency and form. Subtypes are classified based on predominant bowel habits: IBS with constipation, IBS with diarrhea, mixed, or unclassified. IBS is not associated with mucosal inflammation or structural abnormalities.
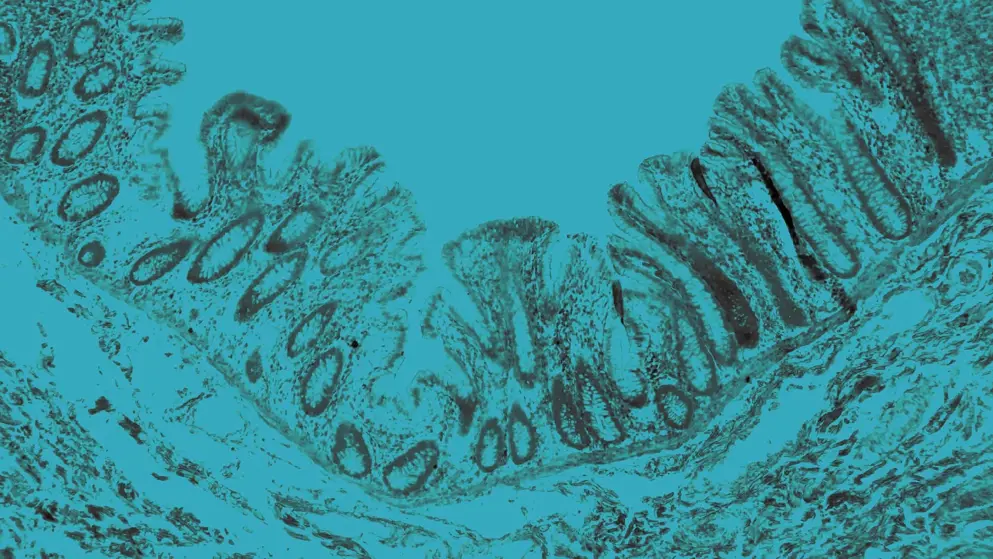
IBD includes Crohn’s disease and ulcerative colitis, both involving chronic, immune-mediated GI inflammation. Crohn’s disease may involve any segment from mouth to anus and is characterized by transmural inflammation and skip lesions. Ulcerative colitis is confined to the colon and rectum, and involves continuous mucosal inflammation.
What are common symptoms of bowel disorders?
Abdominal pain, altered bowel habits, bloating, and fatigue are common to IBS and IBD. However, weight loss, rectal bleeding, and extraintestinal manifestations are more indicative of IBD.
What are key criteria and challenges in diagnosing IBS versus IBD?
IBS is a diagnosis of exclusion, reliant on symptom-based criteria and the absence of biochemical abnormalities. By contrast, IBD diagnosis necessitates a multistep strategy incorporating serologic and fecal biomarkers, cross-sectional imaging, endoscopic evaluation, and histology to confirm inflammation and structural changes in the GI tract.
Despite overlapping symptoms, IBS lacks objective inflammatory markers, while IBD typically presents with detectable mucosal inflammation.
How are bowel disorders managed?
Management is condition-specific. IBS treatment targets symptom relief through dietary changes (e.g., low FODMAP), pharmacologic agents, and behavioral therapies. IBD management includes anti-inflammatories, immunosuppressants, biologics (e.g., anti-TNF, anti-integrin, Janus kinase [JAK] inhibitors) and, in some cases, surgery.
What are risk factors for both IBS and IBD?
Genetic predispositions, stress, diet, and microbiome alterations.
Developed by EPG Health for Medthority, independently of any sponsor.

