
- Home
- /
- Drugs
- /
- B
- /
- Betamethasone Dipropionate
- /
- Betamethasone Dipropionate BETAMETHASONE DIPROPIONATE .5 mg/g Proficient Rx LP
Betamethasone Dipropionate
- Betamethasone Dipropionate BETAMETHASONE DIPROPIONATE .5 mg/g Preferred Pharmaceuticals, Inc.
- Betamethasone Dipropionate BETAMETHASONE DIPROPIONATE .5 mg/g RPK Pharmaceuticals, Inc.
- Betamethasone Dipropionate BETAMETHASONE DIPROPIONATE .5 mg/g Proficient Rx LP
- Betamethasone Dipropionate BETAMETHASONE DIPROPIONATE .5 mg/g REMEDYREPACK INC.
- Betamethasone Dipropionate BETAMETHASONE DIPROPIONATE .5 mg/g E. Fougera & Co. a division of Fougera Pharmaceuticals Inc.
Summary of product characteristics
Adverse Reactions
ADVERSE REACTIONS: The only local adverse reaction reported to be possibly or probably related to treatment with betamethasone dipropionate cream (augmented) during adult, controlled clinical studies was stinging. It occurred in 1 patient, 0.4%, of the 242 patients or subjects involved in the studies. Adverse reactions reported to be possibly or probably related to treatment with betamethasone dipropionate cream (augmented) during a pediatric clinical study include signs of skin atrophy (telangiectasia, bruising, shininess). Skin atrophy occurred in 7 of 67 (10%) patients, involving all age groups from 3 months - 12 years of age. The following local adverse reactions are reported infrequently when topical corticosteroids are used as recommended. These reactions are listed in an approximate decreasing order of occurrence: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, miliaria. Systemic absorption of topical corticosteroids has produced reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing's syndrome, hyperglycemia, and glucosuria in some patients.
Contraindications
CONTRAINDICATIONS: Betamethasone dipropionate cream (augmented), is contraindicated in patients who are hypersensitive to betamethasone dipropionate, to other corticosteroids, or to any ingredient in this preparation.
Description
DESCRIPTION: Betamethasone dipropionate cream (augmented) contains betamethasone dipropionate, USP, a synthetic adrenocorticosteroid, for dermatologic use in an emollient base. Betamethasone, an analog of prednisolone, has a high degree of corticosteroid activity and a slight degree of mineralocorticoid activity. Betamethasone dipropionate is the 17,21-dipropionate ester of betamethasone. Chemically, betamethasone dipropionate is 9-fluoro-11β,17,21-trihydroxy-16β-methylpregna-1,4-diene-3,20-dione 17,21-dipropionate, with the empirical formula C 28 H 37 FO 7 , a molecular weight of 504.6, and the following structural formula: Betamethasone dipropionate is a white to creamy white, odorless crystalline powder, insoluble in water. Each gram of Betamethasone Dipropionate Cream USP (Augmented*), 0.05% contains: 0.643 mg betamethasone dipropionate, USP (equivalent to 0.5 mg betamethasone) in an emollient cream base of purified water, USP; chlorocresol NF; white petrolatum, USP; white wax, NF; cyclomethicone; sorbitol solution, USP; glyceryl monooleate; propylene glycol, USP; ceteareth-30; carbomer 940, NF; and sodium hydroxide NF. Structural Formula
Dosage And Administration
DOSAGE AND ADMINISTRATION: Apply a thin film of betamethasone dipropionate cream (augmented) to the affected skin areas once or twice daily. Treatment with betamethasone dipropionate cream (augmented) should be limited to 45 g per week. Betamethasone dipropionate cream (augmented) is not to be used with occlusive dressings.
Indications And Usage
INDICATIONS AND USAGE: Betamethasone dipropionate cream (augmented) is a high-potency corticosteroid indicated for relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses in patients 13 years and older.
Overdosage
OVERDOSAGE: Topically applied corticosteroids can be absorbed in sufficient amounts to produce systemic effects. (See PRECAUTIONS .)
Clinical Pharmacology
CLINICAL PHARMACOLOGY: The corticosteroids are a class of compounds comprising steroid hormones secreted by the adrenal cortex and their synthetic analogs. In pharmacologic doses, corticosteroids are used primarily for their anti-inflammatory and/or immunosuppressive effects. Topical corticosteroids, such as betamethasone dipropionate, are effective in the treatment of corticosteroid-responsive dermatoses primarily because of their anti-inflammatory, anti-pruritic, and vasoconstrictive actions. However, while the physiologic, pharmacologic, and clinical effects of the corticosteroids are well known, the exact mechanisms of their actions in each disease are uncertain. Betamethasone dipropionate, a corticosteroid, has been shown to have topical (dermatologic) and systemic pharmacologic and metabolic effects characteristic of this class of drugs. Pharmacokinetics: The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings. (See DOSAGE AND ADMINISTRATION section.) Topical corticosteroids can be absorbed through normal intact skin. Inflammation and/or other disease processes in the skin may increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. (See DOSAGE AND ADMINISTRATION section.) Once absorbed through the skin, topical corticosteroids enter pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees, are metabolized primarily in the liver and excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile. Betamethasone dipropionate cream (augmented) was applied once daily at 7 grams per day for 1 week to diseased skin, in adult patients with psoriasis or atopic dermatitis, to study its effects on the hypothalamic-pituitary-adrenal (HPA) axis. The results suggested that the drug caused a slight lowering of adrenal corticosteroid secretion, although in no case did plasma cortisol levels go below the lower limit of the normal range. Sixty-seven pediatric patients ages 1 to 12 years, with atopic dermatitis, were enrolled in an open-label, hypothalamic-pituitary-adrenal (HPA) axis safety study. Betamethasone dipropionate cream (augmented) was applied twice daily for 2 to 3 weeks over a mean body surface area of 58% (range 35% to 95%). In 19 of 60 (32%) evaluable patients, adrenal suppression was indicated by either a ≤ 5 mcg/dL pre-stimulation cortisol, or a cosyntropin post-stimulation cortisol ≤ 18 mcg/dL and/or an increase of < 7 mcg/dL from the baseline cortisol. Studies performed with betamethasone dipropionate cream (augmented) indicate that it is in the high range of potency as compared with other topical corticosteroids.
Pharmacokinetics
Pharmacokinetics: The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings. (See DOSAGE AND ADMINISTRATION section.) Topical corticosteroids can be absorbed through normal intact skin. Inflammation and/or other disease processes in the skin may increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. (See DOSAGE AND ADMINISTRATION section.) Once absorbed through the skin, topical corticosteroids enter pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees, are metabolized primarily in the liver and excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile. Betamethasone dipropionate cream (augmented) was applied once daily at 7 grams per day for 1 week to diseased skin, in adult patients with psoriasis or atopic dermatitis, to study its effects on the hypothalamic-pituitary-adrenal (HPA) axis. The results suggested that the drug caused a slight lowering of adrenal corticosteroid secretion, although in no case did plasma cortisol levels go below the lower limit of the normal range. Sixty-seven pediatric patients ages 1 to 12 years, with atopic dermatitis, were enrolled in an open-label, hypothalamic-pituitary-adrenal (HPA) axis safety study. Betamethasone dipropionate cream (augmented) was applied twice daily for 2 to 3 weeks over a mean body surface area of 58% (range 35% to 95%). In 19 of 60 (32%) evaluable patients, adrenal suppression was indicated by either a ≤ 5 mcg/dL pre-stimulation cortisol, or a cosyntropin post-stimulation cortisol ≤ 18 mcg/dL and/or an increase of < 7 mcg/dL from the baseline cortisol. Studies performed with betamethasone dipropionate cream (augmented) indicate that it is in the high range of potency as compared with other topical corticosteroids.
Effective Time
20210101
Version
3
Spl Product Data Elements
Betamethasone Dipropionate Betamethasone dipropionate BETAMETHASONE DIPROPIONATE BETAMETHASONE WATER CHLOROCRESOL PETROLATUM WHITE WAX CYCLOMETHICONE SORBITOL GLYCERYL OLEATE PROPYLENE GLYCOL CETEARETH-30 CARBOMER HOMOPOLYMER TYPE C (ALLYL PENTAERYTHRITOL CROSSLINKED) SODIUM HYDROXIDE
Carcinogenesis And Mutagenesis And Impairment Of Fertility
Carcinogenesis, Mutagenesis, and Impairment of Fertility: Long-term animal studies have not been performed to evaluate the carcinogenic potential of betamethasone dipropionate. Betamethasone was negative in the bacterial mutagenicity assay ( Salmonella typhimurium and Escherichia coli ), and in the mammalian cell mutagenicity assay (CHO/HGPRT). It was positive in the in-vitro human lymphocyte chromosome aberration assay, and equivocal in the in-vivo mouse bone marrow micronucleus assay. This pattern of response is similar to that of dexamethasone and hydrocortisone. Reproductive studies with betamethasone dipropionate carried out in rabbits at doses of 1.0 mg/kg by the intramuscular route and in mice up to 33 mg/kg by the intramuscular route indicated no impairment of fertility except for dose-related increases in fetal resorption rates in both species. These doses are approximately 5 and 38 fold the human dose based on a mg/m 2 comparison, respectively.
Application Number
ANDA076215
Brand Name
Betamethasone Dipropionate
Generic Name
Betamethasone dipropionate
Product Ndc
63187-286
Product Type
HUMAN PRESCRIPTION DRUG
Route
TOPICAL
Laboratory Tests
Laboratory Tests: The following tests may be helpful in evaluating HPA axis suppression: Urinary free cortisol test ACTH stimulation test
Package Label Principal Display Panel
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – 15 G LABEL NDC 63187-286-15-15 FOUGERA ® Repackaged by: Proficient Rx LP Thousand Oaks, CA 91320 BETAMETHASONE DIPROPIONATE CREAM USP (AUGMENTED*), 0.05% (Potency expressed as betamethasone) *Vehicle augments the penetration of the steroid. Rx only FOR DERMATOLOGIC USE ONLY NOT FOR OPHTHALMIC USE See crimp of tube for Lot Number and Exp. Date. NET WT 15 grams 63187-286-15
Spl Unclassified Section
FOR DERMATOLOGIC USE ONLY NOT FOR OPHTHALMIC USE Rx Only
Information For Patients
Information for Patients: Patients using topical corticosteroids should receive the following information and instructions. This information is intended to aid in the safe and effective use of this medication. It is not a disclosure of all possible adverse or intended effects. • This medication is to be used as directed by the physician and should not be used longer than the prescribed time period. It is for external use only. Avoid contact with the eyes. • Patients should be advised not to use this medication for any disorder other than that for which it was prescribed. • The treated skin area should not be bandaged or otherwise covered or wrapped as to be occlusive. (See DOSAGE AND ADMINISTRATION section.) • Patients should report any signs of local adverse reactions. • Other corticosteroid-containing products should not be used with betamethasone dipropionate cream (augmented) without first talking to your physician.
Nursing Mothers
Nursing Mothers: It is not known whether topical administration of corticosteroids can result in sufficient systemic absorption to produce detectable quantities in breast milk. Systemically administered corticosteroids are secreted into breast milk in quantities not likely to have a deleterious effect on the infant. Nevertheless, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Pediatric Use: Use of betamethasone dipropionate cream (augmented) in pediatric patients 12 years of age and younger is not recommended. (See CLINICAL PHARMACOLOGY and ADVERSE REACTIONS Sections.) In an open label study, 19 of 60 (32%) evaluable pediatric patients (aged 3 months - 12 years old) using betamethasone dipropionate cream (augmented) for treatment of atopic dermatitis demonstrated HPA axis suppression. The proportion of patients with adrenal suppression in this study was progressively greater, the younger the age group. (See CLINICAL PHARMACOLOGY - Pharmacokinetics .) Pediatric patients may demonstrate greater susceptibility to topical corticosteroid-induced HPA axis suppression and Cushing's syndrome than mature patients because of a larger skin surface area to body weight ratio. The study described above supports this premise, as adrenal suppression in 9-12 year olds, 6-8 year olds, 2-5 year olds, and 3 months - 1 year old was 17%, 32%, 38%, and 50% respectively. Hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing's syndrome, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include linear growth retardation, delayed weight gain, low plasma cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema. Chronic corticosteroid therapy may interfere with the growth and development of children.
Pregnancy
Pregnancy: Teratogenic effects: Pregnancy Category C: Corticosteroids are generally teratogenic in laboratory animals when administered systemically at relatively low dosage levels. Betamethasone dipropionate has been shown to be teratogenic in rabbits when given by the intramuscular route at doses of 0.05 mg/kg. This dose is approximately 0.2 fold the maximum human dose based on a mg/m 2 comparison. The abnormalities observed included umbilical hernias, cephalocele and cleft palates. Some corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. There are no adequate and well-controlled studies in pregnant women on teratogenic effects from topically applied corticosteroids. Therefore, topical corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Drugs of this class should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time.
How Supplied
HOW SUPPLIED: Betamethasone Dipropionate Cream USP (Augmented*), 0.05%, is supplied as follows: NDC 63187-286-15 15 gram tube Store between 2° and 30°C (36° and 86°F). E. FOUGERA & CO. A division of Fougera Pharmaceuticals Inc. Melville, New York 11747 Repackaged by: Proficient Rx LP Thousand Oaks, CA 91320 I2265B R09/11 #59
General Precautions
General: Systemic absorption of topical corticosteroids has produced reversible HPA axis suppression, manifestations of Cushing's syndrome, hyperglycemia, and glucosuria in some patients. Conditions which augment systemic absorption include the application of the more potent corticosteroids, use over large surface areas, prolonged use, and the addition of occlusive dressings.Use of more than one corticosteroid-containing product at the same time may increase total systemic glucocorticoid exposure. (See DOSAGE AND ADMINISTRATION section.) Therefore, patients receiving a large dose of a potent topical steroid applied to a large surface area should be evaluated periodically for evidence of HPA axis suppression by using the urinary free cortisol and ACTH stimulation tests. If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid. Recovery of HPA axis function is generally prompt and complete upon discontinuation of the drug. In an open-label pediatric study of 60 evaluable patients, of the 19 who showed evidence of suppression, 4 patients were tested 2 weeks after discontinuation of betamethasone dipropionate cream (augmented), and 3 of the 4 (75%) had complete recovery of HPA axis function. Infrequently, signs and symptoms of steroid withdrawal may occur, requiring supplemental systemic corticosteroids. Children may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity. (See PRECAUTIONS – Pediatric Use .) If irritation develops, topical corticosteroids should be discontinued and appropriate therapy instituted. In the presence of dermatological infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, the corticosteroid should be discontinued until the infection has been adequately controlled.
Precautions
PRECAUTIONS: General: Systemic absorption of topical corticosteroids has produced reversible HPA axis suppression, manifestations of Cushing's syndrome, hyperglycemia, and glucosuria in some patients. Conditions which augment systemic absorption include the application of the more potent corticosteroids, use over large surface areas, prolonged use, and the addition of occlusive dressings.Use of more than one corticosteroid-containing product at the same time may increase total systemic glucocorticoid exposure. (See DOSAGE AND ADMINISTRATION section.) Therefore, patients receiving a large dose of a potent topical steroid applied to a large surface area should be evaluated periodically for evidence of HPA axis suppression by using the urinary free cortisol and ACTH stimulation tests. If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid. Recovery of HPA axis function is generally prompt and complete upon discontinuation of the drug. In an open-label pediatric study of 60 evaluable patients, of the 19 who showed evidence of suppression, 4 patients were tested 2 weeks after discontinuation of betamethasone dipropionate cream (augmented), and 3 of the 4 (75%) had complete recovery of HPA axis function. Infrequently, signs and symptoms of steroid withdrawal may occur, requiring supplemental systemic corticosteroids. Children may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity. (See PRECAUTIONS – Pediatric Use .) If irritation develops, topical corticosteroids should be discontinued and appropriate therapy instituted. In the presence of dermatological infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, the corticosteroid should be discontinued until the infection has been adequately controlled. Information for Patients: Patients using topical corticosteroids should receive the following information and instructions. This information is intended to aid in the safe and effective use of this medication. It is not a disclosure of all possible adverse or intended effects. • This medication is to be used as directed by the physician and should not be used longer than the prescribed time period. It is for external use only. Avoid contact with the eyes. • Patients should be advised not to use this medication for any disorder other than that for which it was prescribed. • The treated skin area should not be bandaged or otherwise covered or wrapped as to be occlusive. (See DOSAGE AND ADMINISTRATION section.) • Patients should report any signs of local adverse reactions. • Other corticosteroid-containing products should not be used with betamethasone dipropionate cream (augmented) without first talking to your physician. Laboratory Tests: The following tests may be helpful in evaluating HPA axis suppression: Urinary free cortisol test ACTH stimulation test Carcinogenesis, Mutagenesis, and Impairment of Fertility: Long-term animal studies have not been performed to evaluate the carcinogenic potential of betamethasone dipropionate. Betamethasone was negative in the bacterial mutagenicity assay ( Salmonella typhimurium and Escherichia coli ), and in the mammalian cell mutagenicity assay (CHO/HGPRT). It was positive in the in-vitro human lymphocyte chromosome aberration assay, and equivocal in the in-vivo mouse bone marrow micronucleus assay. This pattern of response is similar to that of dexamethasone and hydrocortisone. Reproductive studies with betamethasone dipropionate carried out in rabbits at doses of 1.0 mg/kg by the intramuscular route and in mice up to 33 mg/kg by the intramuscular route indicated no impairment of fertility except for dose-related increases in fetal resorption rates in both species. These doses are approximately 5 and 38 fold the human dose based on a mg/m 2 comparison, respectively. Pregnancy: Teratogenic effects: Pregnancy Category C: Corticosteroids are generally teratogenic in laboratory animals when administered systemically at relatively low dosage levels. Betamethasone dipropionate has been shown to be teratogenic in rabbits when given by the intramuscular route at doses of 0.05 mg/kg. This dose is approximately 0.2 fold the maximum human dose based on a mg/m 2 comparison. The abnormalities observed included umbilical hernias, cephalocele and cleft palates. Some corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. There are no adequate and well-controlled studies in pregnant women on teratogenic effects from topically applied corticosteroids. Therefore, topical corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Drugs of this class should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time. Nursing Mothers: It is not known whether topical administration of corticosteroids can result in sufficient systemic absorption to produce detectable quantities in breast milk. Systemically administered corticosteroids are secreted into breast milk in quantities not likely to have a deleterious effect on the infant. Nevertheless, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. Pediatric Use: Use of betamethasone dipropionate cream (augmented) in pediatric patients 12 years of age and younger is not recommended. (See CLINICAL PHARMACOLOGY and ADVERSE REACTIONS Sections.) In an open label study, 19 of 60 (32%) evaluable pediatric patients (aged 3 months - 12 years old) using betamethasone dipropionate cream (augmented) for treatment of atopic dermatitis demonstrated HPA axis suppression. The proportion of patients with adrenal suppression in this study was progressively greater, the younger the age group. (See CLINICAL PHARMACOLOGY - Pharmacokinetics .) Pediatric patients may demonstrate greater susceptibility to topical corticosteroid-induced HPA axis suppression and Cushing's syndrome than mature patients because of a larger skin surface area to body weight ratio. The study described above supports this premise, as adrenal suppression in 9-12 year olds, 6-8 year olds, 2-5 year olds, and 3 months - 1 year old was 17%, 32%, 38%, and 50% respectively. Hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing's syndrome, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include linear growth retardation, delayed weight gain, low plasma cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema. Chronic corticosteroid therapy may interfere with the growth and development of children.
Relevant Learning Zones
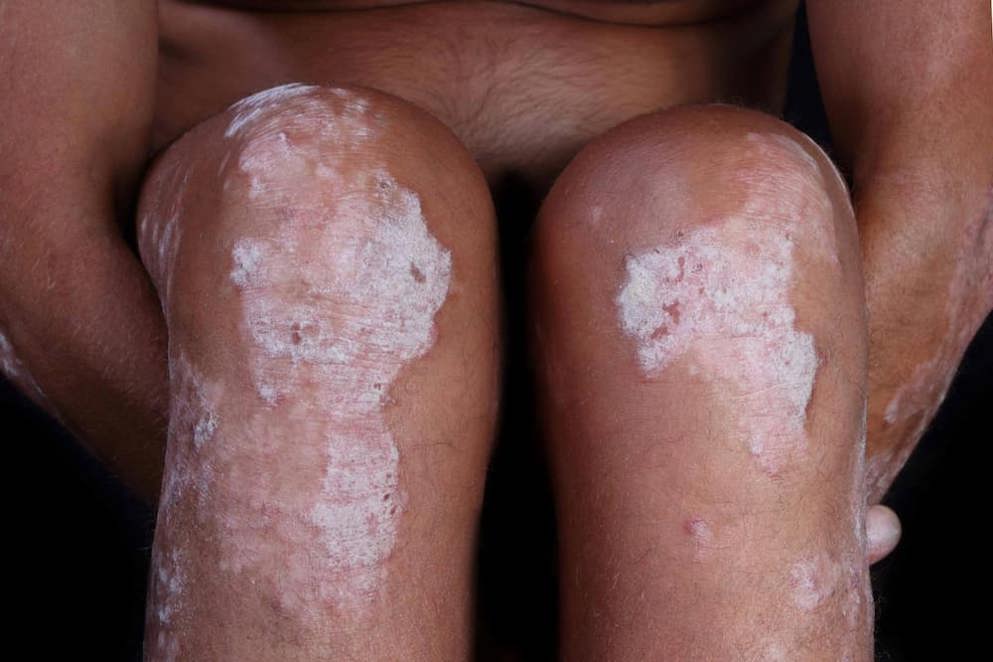
When it comes to psoriasis, a knowledgeable healthcare professional is highly valued by patients (Pariser et al., 2016). With this in mind, we created The Psoriasis Academy, an online hub featuring succinct and easily accessible information for all healthcare professionals who want to know more about psoriasis and keep up to date with the latest developments.

This Learning Zone introduces you to our young patient Mia who, along with our paediatric psoriasis experts, helps us to describe the burden of childhood psoriasis for patients and their families, particularly the difficulties around diagnosis, comorbidities and limited treatment options.
Learning Zones
The Learning Zones are an educational resource for healthcare professionals that provide medical information on the epidemiology, pathophysiology and burden of disease, as well as diagnostic techniques and treatment regimens.
Disclaimer
The drug Prescribing Information (PI), including indications, contra-indications, interactions, etc, has been developed using the U.S. Food & Drug Administration (FDA) as a source (www.fda.gov).
Medthority offers the whole library of PI documents from the FDA. Medthority will not be held liable for explicit or implicit errors, or missing data.
Drugs appearing in this section are approved by the FDA. For regions outside of the United States, this content is for informational purposes only and may not be aligned with local regulatory approvals or guidance.
