News
FYB 201, a biosimilar to Lucentis, is filed at the FDA.
Formycon AG and its license partner Bioeq AG announce that the biologics license application (BLA) for FYB 201, Formycon’s biosimilar candidate to Lucentis has been recently submitted to the FDA.
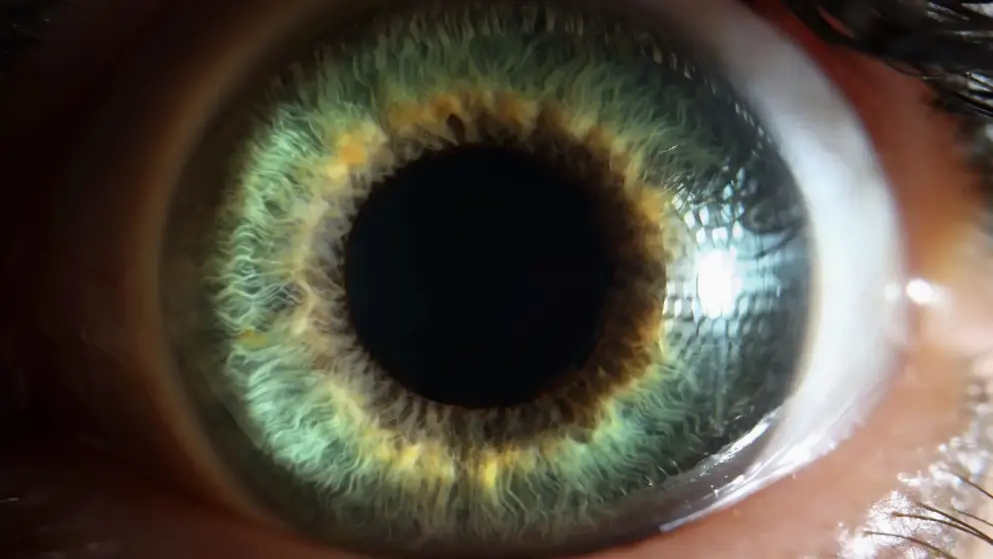
Lucentis is used in the treatment of neovascular (wet) macular degeneration and other serious eye diseases. It inhibits vascular endothelial growth factor (VEGF), which is responsible for the excessive formation of blood vessels in the retina.
If approved, Coherus BioSciences, Inc. will commercialize FYB 201 (also known as CHS-201) in the United States of America per the terms of the November 2019 license and development agreement between Bioeq and Coherus. Bioeq is a Swiss biopharmaceutical joint venture between the Polpharma Biologics Group and the Strüngmann Group and develops, licenses and commercializes biosimilars. Coherus is a U.S. pharmaceutical company with an expertise in biosimilar development and commercialization.
Condition: Age Related Macular Degeneration
Type: drug