News
CHMP recommends approval of Entyvio for ulcerative colitis or Crohn’s disease.- Takeda Pharma
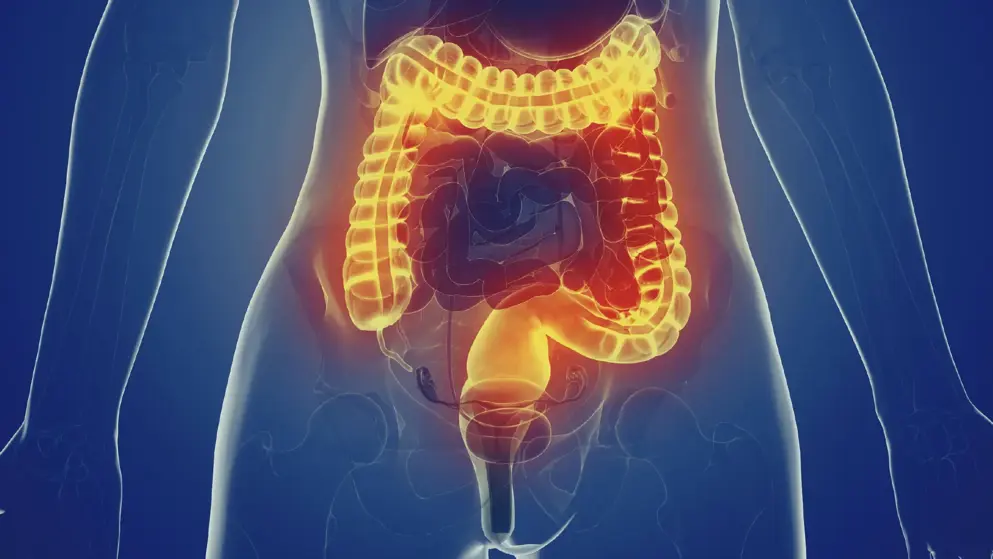
Takeda Pharmaceutical Co announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has recommended the approval of a subcutaneous (SC) formulation of the gut-selective biologic Entyvio (vedolizumab) for use as maintenance therapy in adults with moderately to severely active ulcerative colitis (UC) or Crohn’s disease (CD). Takeda proposes to make vedolizumab SC available in both a pre-filled syringe and a pre-filled pen. The positive opinion from the CHMP was based on the pivotal phase III VISIBLE trials which assessed the safety and efficacy of a SC formulation of vedolizumab as maintenance therapy in adult patients with moderately to severely active UC or CD who achieved clinical response at week 6 following two doses of open-label vedolizumab intravenous (IV) therapy at weeks 0 and 2. Data from a long-term, open-label extension study of patients from VISIBLE 1 and VISIBLE 2 was also considered.
Condition: Ulcerative Colitis
Type: drug