
Understanding Crohn’s disease
Get to grips with Crohn’s disease, an increasingly common and debilitating form of IBD.
- Discover how early intervention could slow or even prevent disease progression
- Identify common symptoms experienced by patients with this condition
- Check out the global incidence with our insightful world map
In this section
Epidemiology
Prevalence and incidence
Crohn’s disease is an inflammatory bowel disease (IBD), characterised by chronic inflammation which can occur at any point along the gastrointestinal tract1. A growing body of evidence suggests that the prevalence and incidence of this debilitating condition is increasing globally, with some regional variation2–5.
While prevalence describes actual numbers of cases, incidence measures rates of new cases, and most epidemiological studies suggest that the incidence of IBD is increasing6.
Until recently, IBD was widely regarded as a disease of the western world4. The prevalence of IBD in westernised countries increased considerably over the course of the 20th century and today exceeds 0.3% in many regions of Europe, North America, and Oceania4. Interestingly, although this figure now appears to be stabilising or even decreasing in some of these countries, the beginning of the 21st century has seen a similar increase in the number of cases reported in newly industrialised nations, with incidence of IBD on the rise in South America, eastern Europe, Asia, and Africa (Figure 1)4. In Brazil, for example, an annual percentage increase in incidence of Crohn’s disease of 11.1% (95% confidence interval 4.8–17.8) has been reported 4.
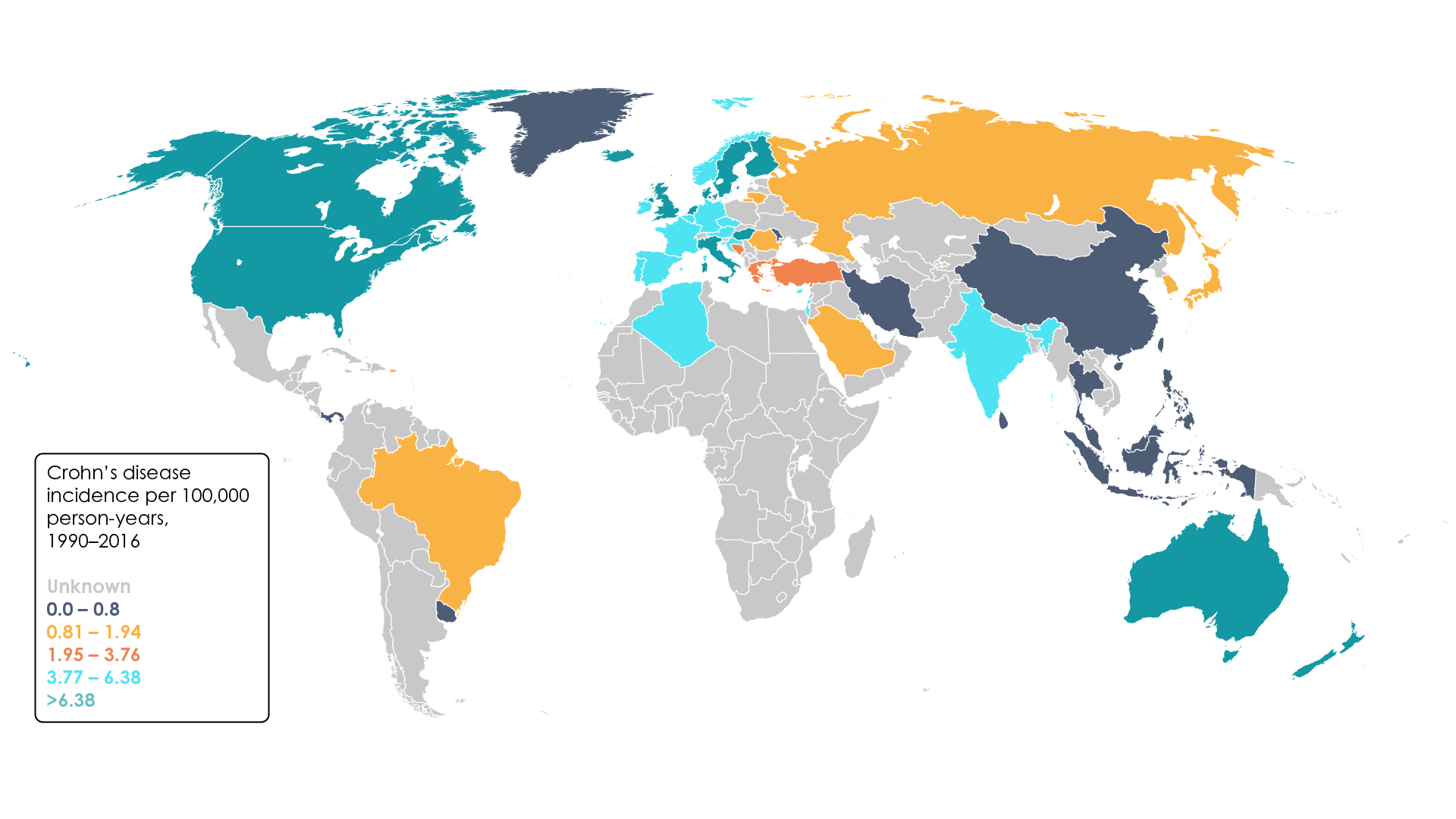
Figure 1. Worldwide incidence of Crohn’s disease per 100,000 person years, 1990–2016 (Adapted4).
Nevertheless, the results of a large systematic review suggest that the prevalence of Crohn’s disease remains highest in Europe (322 cases per 100,000 individuals in Germany) and North America (319 cases per 100,000 individuals in Canada)4,7. More recently, similar findings were reported by a study based on data from the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD)2. This revealed that between 1990 and 2017 the highest age-standardised prevalence rate of IBD was recorded in high-income North America (344.8 cases per 100,000 individuals [95% UI 331.7 – 359.3] in 1990; 422.0 cases per 100,000 individuals [95% UI 398.7 – 466.1] in 2017)2. Meanwhile, the lowest age-standardised prevalence rates were reported in the Caribbean (6.7 cases per 100,000 individuals [95% UI 6.3 – 7.2] in 2017)2. Other regions with low rates of IBD included Andean Latin America and parts of sub-Saharan Africa2.
Disease development
Among adults, Crohn’s disease affects more women than men8,9. It can develop at any point from early childhood but the median age of onset is 30 years6,10. Onset tends to occur in two peaks – mainly between 20 and 30 years of age, but also around 50 years10.
Aetiology
Genetic susceptibility
Though the exact aetiology of Crohn’s disease remains unknown, familial aggregation studies and twin studies indicate a strong genetic component11,12. A family history of the condition has been reported by approximately 12% of patients and concordance rates among monozygotic twins are estimated to be around 50%1,3.
Further evidence of a genetic component can be found in differences in the prevalence of Crohn’s disease between ethnic groups. Caucasians and African Americans are among those most commonly affected, while individuals of Hispanic and Asian descent are considered to be at lower risk11. Of note, risk of developing Crohn’s disease is reportedly 3–fold higher in Jewish compared with non-Jewish individuals12.
To date, genome-wide association studies have identified over 200 susceptibility loci for IBD13. In Crohn’s disease, these include polymorphisms in the NOD2 gene, which is expressed in a range of cell types and encodes a protein with a role in innate immunity, as well as in autophagy-related genes14–16.
Gut flora
Dysbiosis is thought to play a key role in IBD pathogenesis, and Crohn’s disease is associated with the presence of specific pathogens in the gut microbiota and a reduction in gut microbial diversity compared to healthy individuals17. Alterations in the composition of the intestinal microbiota in patients with Crohn’s disease include changes in the relative abundance of Bacteroides and Firmicutes, and an increased proportion of Gammaproteobacteria and Enterobacteriales, as well as a decrease in Clostridiales17.
Environmental factors
A range of environmental factors influence the onset and progression of Crohn’s disease in genetically susceptible individuals1.
Cigarette smoking is the best-studied environmental risk factor. Among smokers, risk of developing Crohn’s disease is reportedly double that of non-smokers and smoking has been linked to an earlier onset of disease, need for immunosuppression, increased need for surgical intervention and higher rates of postoperative recurrence1,3.
Dietary changes and associated gut dysbiosis has also been implicated in the development of Crohn’s disease1. While high fibre diets are thought to reduce risk of Crohn’s disease, diets rich in sugar, fat, and meat have been shown to increase risk in several studies10.
Crohn’s disease risk is also held to be increased by exposure to certain drugs, including oral contraceptives, aspirin and non-steroidal anti-inflammatory drugs (NSAIDs), as well as use of antibiotics such as penicillins, cephalosporins, metronidazole and fluoroquinolones, particularly during childhood1,10,18.
Pathophysiology
Pathogenesis
The pathogenesis of Crohn’s disease is not well understood, involving inflammatory processes of the intestinal tract, the origins of which are unknown19,20. This being said, it is thought that both the innate and adaptive immune responses play a role21. Indeed, massive infiltration of the mucosa by innate and adaptive immune cells is considered a key characteristic of active IBD in general22. This may be related to either overactivation of effector T cells and/or alteration of T cell-mediated tolerance22. An abnormal and sustained host immune response against intestinal microbiota is key to the development of IBD22.
In patients with Crohn’s disease, damage to the intestinal barrier leads to translocation of commensal microorganisms into the bowel wall. Innate immune cells respond to microbes and their mediators, resulting in immune cell activation and the production of cytokines and chemokines. Innate immunity is then activated as a result of disruptions to gut homeostasis and recruitment of additional immune cells to the bowel wall and causes activation of adaptive immune cells. Activated immune cells produce mediators, including cytokines, which, along with immune cells themselves, cause damage to epithelial cells, impair barrier function resulting in dysbiosis, and perpetuate gut inflammation (Figure 2)20 .
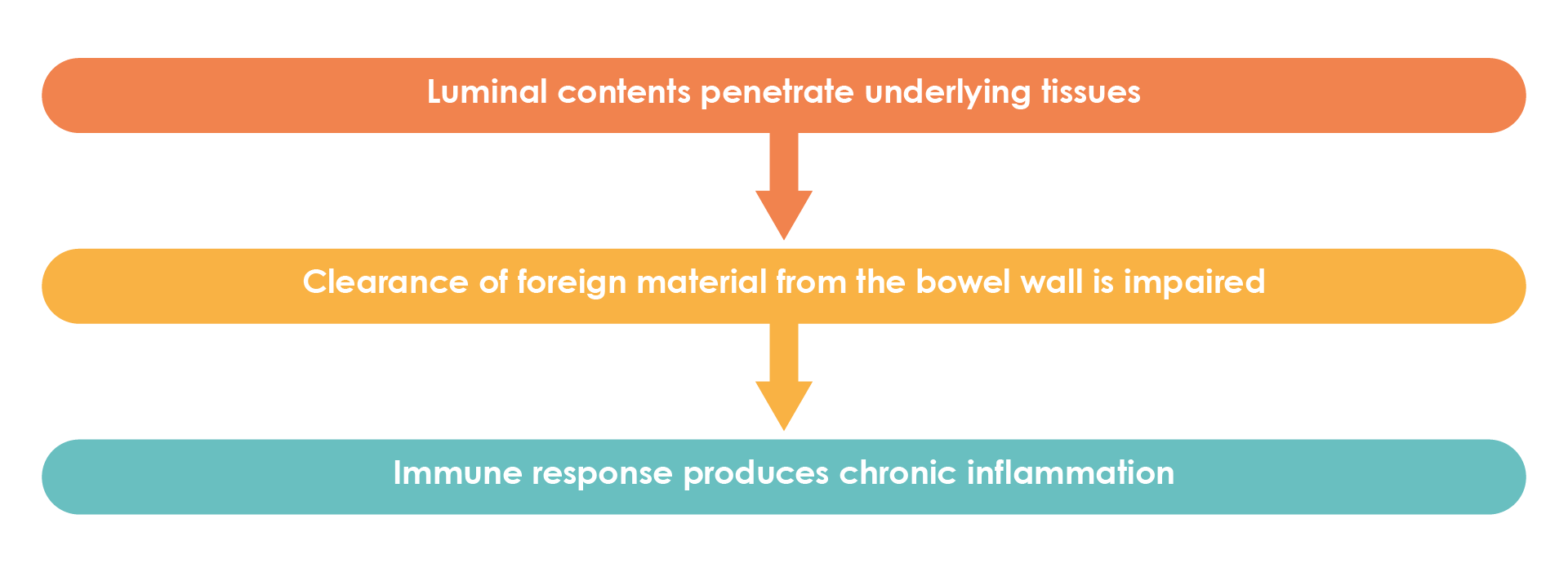
Figure 2. Pathogenesis of Crohn’s disease (Adapted23).
Crohn’s disease is a chronic progressive disease, with half of all patients developing intestinal complications, including strictures and fistulae, within 10 years of diagnosis1.
Crohn’s disease is characterised by patchy, transmural inflammation, known as “skip lesions”1,20. The lesions that typically occur in Crohn’s disease are “cobblestone appearance” and “longitudinal ulcers”24. It can occur at any point along the gastrointestinal tract, where it causes chronic, relapsing transmural inflammation, which is associated with a range of debilitating symptoms including diarrhoea, abdominal pain, fever, and fatigue1,10,20.
At diagnosis, an estimated 81% of patients with Crohn’s disease present with a luminal inflammatory phenotype, though this may change as the disease progresses10,25,26. Extraintestinal manifestations of the disease may impact patient quality of life, long-term outcomes, risk of hospitalisation, complications and surgery1. Numerous factors are thought to affect quality of life in patients with Crohn’s disease, including1:
- Physical status and ability to function (e.g. pain, defecation, sleep loss, fatigue)
- Psychological status and wellbeing (e.g. body image, emotional status)
- Economic and/or vocational factors (e.g. ability to participate in educational/recreational activities or to work and earn money)
- Social factors (e.g. relationships and sexual function)
Treatment goals
Evolution of treatment goals and treat-to-target
In the past, standard of care for patients with IBD was limited to managing their symptoms. Today, treatment goals in Crohn’s disease are evolving and a treat-to-target approach is now encouraged. This strategy promotes personalised care and early intervention27.
A treat-to-target approach involves careful monitoring of specific and objective improvement in measures of inflammation27,28.
Currently mucosal healing represents an important treatment target in Crohn’s disease and has been associated with improved patient outcomes1. In the future, transmural healing, which can be assessed using cross-sectional imaging techniques, is expected to become another key treatment target for patients with Crohn’s disease (Figure 3)1,29.
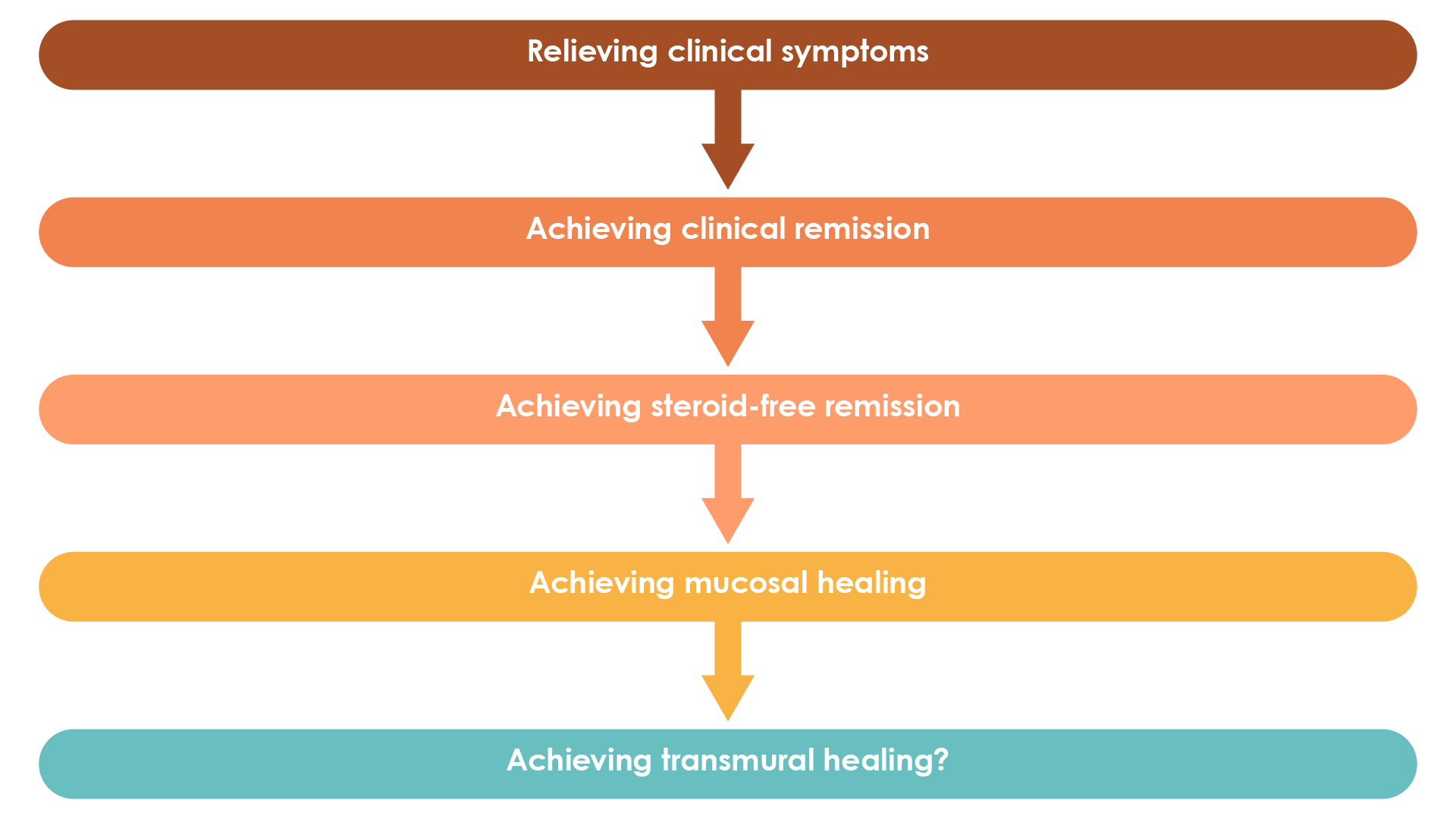
Figure 3. Evolution of treatment targets in Crohn’s disease (Adapted30).
Consensus definition of targets in Crohn’s disease: STRIDE
The Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) programme assessed potential treatment targets for patients with IBD to be used in a treat-to-target strategy31. For Crohn’s disease, the expert-consensus definition of treatment targets that was generated by STRIDE and published in 2015, combines clinical (patient-reported) remission and endoscopic (clinician-reported) remission31,32.
The original STRIDE statements define clinical remission as resolution of abdominal pain and normalisation of bowel habit, while absence of ulceration at ileocolonoscopy is used to confirm endoscopic remission31.
More recently, an updated version of the original STRIDE statements, STRIDE-II, has been made available28. Clinical response and remission, endoscopic healing, and normalisation of C-reactive protein/erythrocyte sedimentation rate and calprotectin were all listed as key therapeutic targets28.
As well as confirming the long-term treatment targets of clinical remission and endoscopic healing that were outlined in STRIDE I, STRIDE-II also introduced several additional targets including absence of disability, restoration of quality of life and normal growth in children28. Symptomatic relief and normalisation of serum and faecal markers were also identified as short-term targets28.
Role of early intervention
Although the majority of patients with Crohn’s disease initially present with uncomplicated disease, for many, chronic intestinal inflammation ultimately leads to the development of irreversible bowel damage and complications, which could necessitate surgery, leading to further permanent bowel damage3,20,33. Nevertheless, experts have now identified a “window of opportunity” in the early stages of the disease, during which appropriate therapeutic intervention has the potential to stop the inflammatory cascade and alter disease progression in many patients32,34.
It is therefore thought that early intervention with effective agents may allow many patients to achieve complete disease control by resolving local inflammation and potentially preventing disease progression and the associated irreversible bowel damage33,35–37.
The potential benefits of early intervention with biological therapies have been highlighted in several publications, including a recent systematic review and meta-analysis in which a total of 18,471 patients were studied38. This study reported that, compared with later biological intervention, early initiation of biological therapy was associated with higher rates of clinical remission (OR 2.10 [95% CI:1.69 – 2.60], n = 2763, p<0.00001) and mucosal healing (OR 2.37 [95% CI: 1.78 – 3.16], n=994, P<0.00001), as well as lower rates of relapse (OR 0.31 [95% CI: 0.14 – 0.68], n=596, p=0.003)38.
Similarly, in another study, higher rates of clinical remission were observed in patients who received early treatment with adalimumab (within 1 year of diagnosis) (66.25%) compared with patients for whom initiation of adalimumab treatment was delayed for 1 year or more (33.77%) (p<0.001)39. In total, 53.75% of patients receiving early adalimumab treatment achieved mucosal healing, compared to 20.78% of those whose treatment was delayed (p<0.001)39. Dose escalation was also required less frequently in patients receiving early (30.00%) compared to late (66.23%) treatment (p<0.01) and the proportion of non-responders was 7.50% and 22.08% respectively (p<0.01)39.
Further insight was provided by a study which reported a significantly lower Lémann Index (a measure of cumulative bowel damage in patients with Crohn’s disease) in patients who were treated with an anti-TNF agent within two years of diagnosis compared to those whose treatment was delayed beyond 2 years (p=0.015)40.
The Lémann Index is used to assess cumulative structural bowel damage in patients with Crohn’s disease40,41.
Nevertheless, in a study that included 16,260 patients with Crohn’s disease, it was reported that less than 5% were initially treated with biologics42. Although conventional therapy is only effective in approximately 20–40% of patients with IBD, the most common initial treatment for Crohn’s disease were found to be corticosteroid monotherapy (42%) and 5-aminosalicylic acid monotherapy (35%)42.
Role of mucosal healing
Recognition of the fact that disease activity often persists in the absence of clinical manifestations highlighted a need for more stringent treatment targets for patients with Crohn’s disease, including mucosal healing30.
Achieving mucosal healing has several important benefits for patients with Crohn’s disease and is now widely considered to be an important therapeutic endpoint. Mucosal healing is associated with sustained clinical remission and a reduced risk of both hospitalisation and surgery30,43.
Nevertheless, it should be noted that given the transmural nature of Crohn’s diseases, there is some debate as to whether or not mucosal healing is an adequate measure of the inflammatory burden and prognosis in patients with this condition. This highlights the need for further investigation of transmural healing as a potential therapeutic target44. Currently available research suggests that transmural healing may represent a deeper level of healing than mucosal healing29. It is hoped that stronger therapeutic intervention earlier in the disease course could lead to increased rates of transmural healing among patients with Crohn’s disease29. The reported benefits of achieving transmural include lower rates of hospitalisation and surgery, as well as an association with favourable longer-term outcomes29.
References
- Roda G, Chien Ng S, Kotze PG, Argollo M, Panaccione R, Spinelli A, et al. Crohn’s disease. Nat Rev Dis Prim. 2020;6(1). doi:10.1038/s41572-020-0156-2.
- Alatab S, Sepanlou SG, Ikuta K, Vahedi H, Bisignano C, Safiri S, et al. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5(1):17–30.
- Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L. Crohn’s disease. Lancet. 2017;389(10080):1741–1755.
- Ng SC, Shi HY, Hamidi N, Underwood FE, Tang W, Benchimol EI, et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2017;390(10114):2769–2778.
- Kaplan GG. The global burden of IBD: From 2015 to 2025. Nat Rev Gastroenterol Hepatol. 2015;12(12):720–727.
- Wehkamp J, Götz M, Herrlinger K, Steurer W, Stange EF. Inflammatory bowel disease. Dtsch Arztebl Int. 2016;113(5):72–82.
- Molodecky NA, Soon IS, Rabi DM, Ghali WA, Ferris M, Chernoff G, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142(1):46-54.e42.
- Adams SM, Bornemann PH. Ulcerative colitis. Am Fam Physician. 2013;87(10):699–705.
- Shah SC, Khalili H, Gower-Rousseau C, Olen O, Benchimol EI, Lynge E, et al. Sex-Based Differences in Incidence of Inflammatory Bowel Diseases—Pooled Analysis of Population-Based Studies From Western Countries. Gastroenterology. 2018;155(4):1079-1089.e3.
- Feuerstein JD, Cheifetz AS. Crohn Disease: Epidemiology, Diagnosis, and Management. Mayo Clin Proc. 2017;92(7):1088–1103.
- Gajendran M, Loganathan P, Catinella AP, Hashash JG. A comprehensive review and update on Crohn’s disease. Dis Mon. 2018;64(2):20–57.
- Ananthakrishnan AN. Epidemiology and risk factors for IBD. Nat Rev Gastroenterol Hepatol. 2015;12(4):205–217.
- de Lange KM, Moutsianas L, Lee JC, Lamb CA, Luo Y, Kennedy NA, et al. Genome-wide association study implicates immune activation of multiple integrin genes in inflammatory bowel disease. Nat Genet. 2017;49(2):256–261.
- Chen Y, Salem M, Boyd M, Bornholdt J, Li Y, Coskun M, et al. Relation between NOD2 genotype and changes in innate signaling in Crohn’s disease on mRNA and miRNA levels. npj Genomic Med. 2017;2(1):1–11.
- Negroni A, Pierdomenico M, Cucchiara S, Stronati L. NOD2 and inflammation: Current insights. J Inflamm Res. 2018;11:49–60.
- Kim S, Eun HS, Jo E-K. Roles of Autophagy-Related Genes in the Pathogenesis of Inflammatory Bowel Disease. Cells. 2019;8(1):77.
- Magro DO, Santos A, Guadagnini D, de Godoy FM, Silva SHM, Lemos WJF, et al. Remission in Crohn’s disease is accompanied by alterations in the gut microbiota and mucins production. Sci Rep. 2019;9(1):1–10.
- Theochari NA, Stefanopoulos A, Mylonas KS, Economopoulos KP. Antibiotics exposure and risk of inflammatory bowel disease: a systematic review. Scand J Gastroenterol. 2018;53(1):1–7.
- Silva FAR, Rodrigues BL, de Lourdes S Ayrizono M, Leal RF. The Immunological Basis of Inflammatory Bowel Disease. Gastroenterol Res Pract. 2016;2097274.
- Neurath MF. Targeting immune cell circuits and trafficking in inflammatory bowel disease. Nat Immunol. 2019;20(8):970–979.
- Aggeletopoulou I, Assimakopoulos SF, Konstantakis C, Triantos C. Interleukin 12/interleukin 23 pathway: Biological basis and therapeutic effect in patients with Crohn’s disease. World J Gastroenterol. 2018;24(36):4093–4103.
- Larmonier CB, Shehab KW, Ghishan FK, Kiela PR. T Lymphocyte Dynamics in Inflammatory Bowel Diseases: Role of the Microbiome. Biomed Res Int. 2015;2015:504638.
- Matricon J, Barnich N, Ardid D. Immunopathogenesis of inflammatory bowel disease. Self Nonself. 2010;1(4):299–309.
- Hisamatsu T. Typical endoscopic findings and diagnostic criteria for Crohn’s disease. 2017. Tokyo. Springer Japan https://link.springer.com/book/10.1007/978-4-431-56018-0.
- Nguyen NH, Singh S, Sandborn WJ. Positioning Therapies in the Management of Crohn’s Disease. Clin Gastroenterol Hepatol. 2020;18(6):1268–1279.
- Thia KT, Sandborn WJ, Harmsen WS, Zinsmeister AR, Loftus EV. Risk factors associated with progression to intestinal complications of Crohn’s disease in a population-based cohort. Gastroenterology. 2010;139(4):1147–1155.
- Drescher H, Lissoos T, Hajisafari E, Evans ER. Treat-to-Target Approach in Inflammatory Bowel Disease: The Role of Advanced Practice Providers. J Nurse Pract. 2019;15(9):676–681.
- Turner D, Ricciuto A, Lewis A, D’Amico F, Dhaliwal J, Griffiths AM, et al. STRIDE-II: An Update on the Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) Initiative of the International Organization for the Study of IBD (IOIBD): Determining Therapeutic Goals for Treat-to-Target strategies in IBD. Gastroenterology. 2021;160(5):1570–1583.
- Serban ED. Treat-to-target in Crohn’s disease: Will transmural healing become a therapeutic endpoint? World J Clin Cases. 2018;6(12):501–513.
- Klenske E, Bojarski C, Waldner M, Rath T, Neurath MF, Atreya R. Targeting mucosal healing in Crohn’s disease: what the clinician needs to know. Ther Adv Gastroenterol. 2019;12:1756284819856865.
- Peyrin-Biroulet L, Sandborn W, Sands BE, Reinisch W, Bemelman W, Bryant R V., et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): Determining Therapeutic Goals for Treat-to-Target. Am J Gastroenterol. 2015;110(9):1324–1338.
- Colombel JF, Narula N, Peyrin-Biroulet L. Management Strategies to Improve Outcomes of Patients With Inflammatory Bowel Diseases. Gastroenterology. 2017;152(2):351–361.
- Danese S, Fiorino G, Peyrin-Biroulet L. Early intervention in Crohn’s disease: Towards disease modification trials. Gut. 2017;66(12):2179–2187.
- Pariente B, Cosnes J, Danese S, Sandborn WJ, Lewin M, Fletcher JG, et al. Development of the Crohn’s disease digestive damage score, the Lémann score. Inflamm Bowel Dis. 2011;17(6):1415–1422.
- Berg DR, Colombel JF, Ungaro R. The role of early biologic therapy in inflammatory bowel disease. Inflamm Bowel Dis. 2019;25(12):1896–1905.
- Feagan BG, Lasch K, Lissoos T, Cao C, Wojtowicz AM, Khalid JM, et al. Rapid Response to Vedolizumab Therapy in Biologic-Naive Patients With Inflammatory Bowel Diseases. Clin Gastroenterol Hepatol. 2019;17(1):130-138.e7.
- Patel H, Latremouille-Viau D, Burne R, Shi S, Adsul S. Comparison of Real-World Treatment Outcomes With Vedolizumab Versus Infliximab in Biologic-Naive Patients With Inflammatory Bowel Disease. Crohns Colitis 360. 2019;1(2). doi:10.1093/crocol/otz022.
- Ungaro RC, Aggarwal S, Topaloglu O, Lee WJ, Clark R, Colombel JF. Systematic review and meta-analysis: efficacy and safety of early biologic treatment in adult and paediatric patients with Crohn’s disease. Aliment Pharmacol Ther. 2020;51(9):831–842.
- Mastronardi M, Curlo M, Cavalcanti E, Burattini O, Cuppone R, Tauro R, et al. Administration Timing Is the Best Clinical Outcome Predictor for Adalimumab Administration in Crohn’s Disease. Front Med. 2019;6:234.
- Lauriot Dit Prevost C, Azahaf M, Nachury M, Branche J, Gerard R, Wils P, et al. Bowel damage and disability in Crohn’s disease: a prospective study in a tertiary referral centre of the Lémann Index and Inflammatory Bowel Disease Disability Index. Aliment Pharmacol Ther. 2020;51(9):889–898.
- Pariente B, Mary JY, Danese S, Chowers Y, De Cruz P, D’Haens G, et al. Development of the Lémann index to assess digestive tract damage in patients with Crohn’s disease. Gastroenterology. 2015;148(1):52-63.e3.
- Siegel CA, Yang F, Eslava S, Cai Z. Treatment Pathways Leading to Biologic Therapies for Ulcerative Colitis and Crohnʼs Disease in the United States. Clin Transl Gastroenterol. 2020;11(2):e00128.
- Picco MF, Farraye FA. Targeting mucosal healing in Crohn’s disease. Gastroenterol Hepatol. 2019;15(10):529–538.
- Fernandes SR, Rodrigues R V., Bernardo S, Cortez-Pinto J, Rosa I, da Silva JP, et al. Transmural Healing Is Associated with Improved Long-term Outcomes of Patients with Crohn’s Disease. Inflamm Bowel Dis. 2017;23(8):1403–1409.
of interest
are looking at
saved
next event
Developed by EPG Health for Medthority in collaboration with Takeda, with some content provided by Takeda.
C-ANPROM/INT/IBDD/0041 October 2021.

